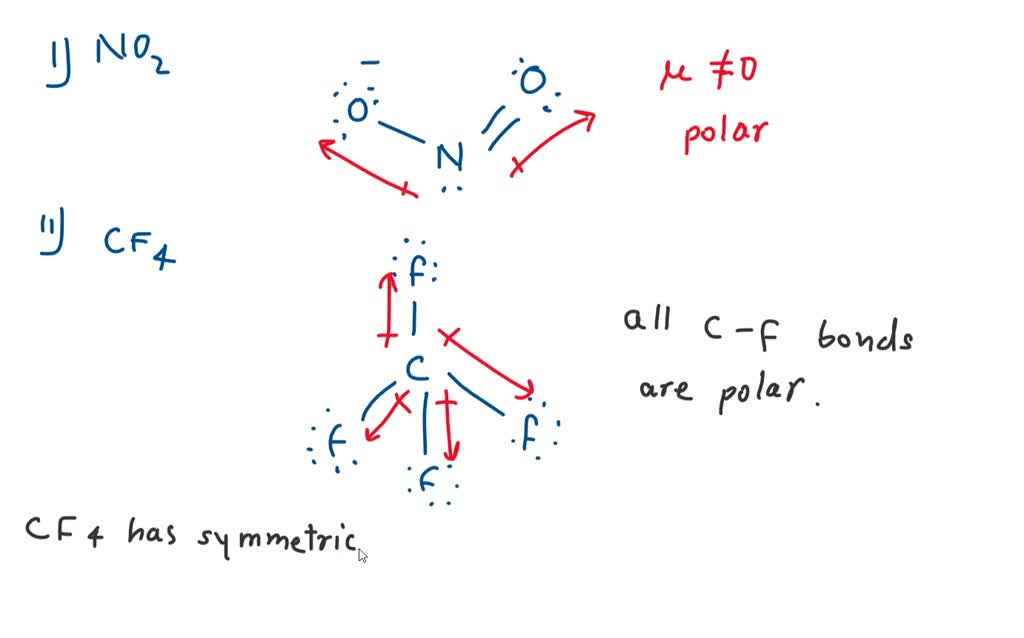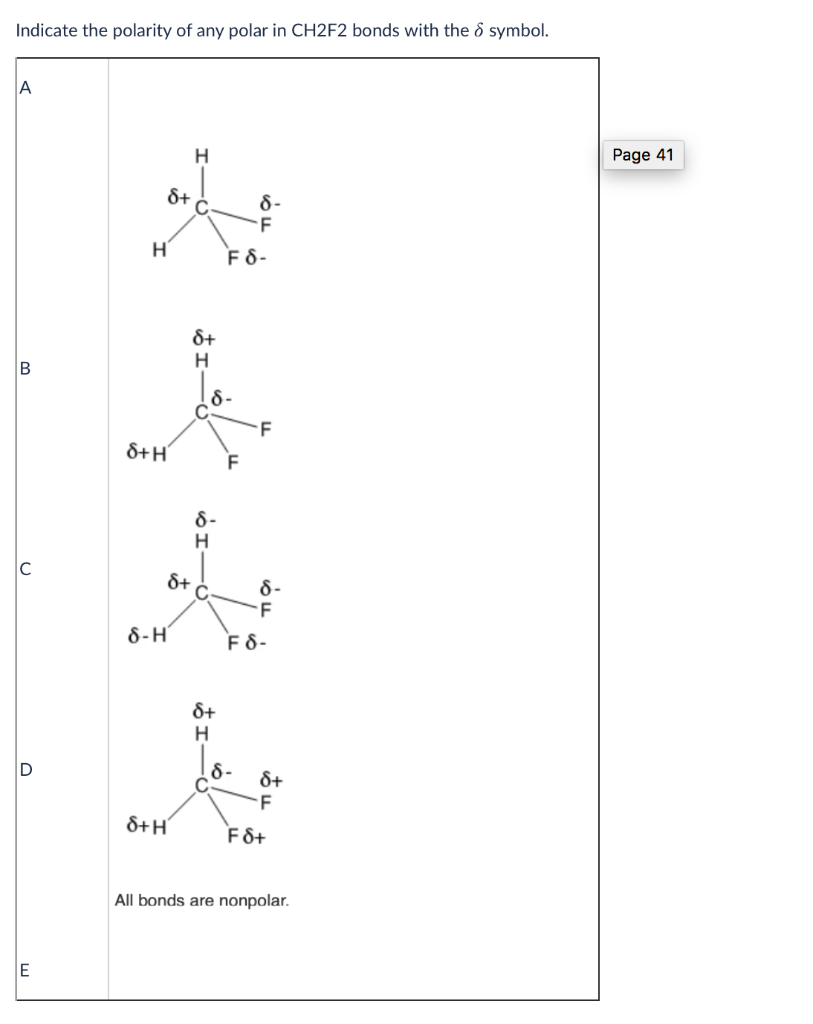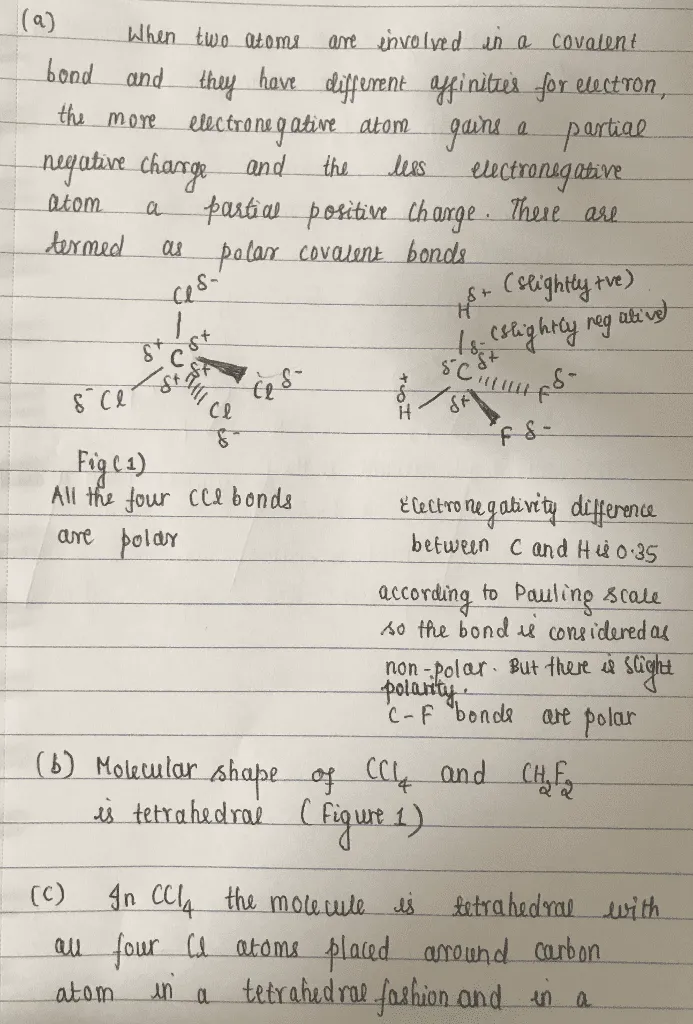
OneClass: -label the polar bonds in each molecule with its correct molecular shape, and then decide i...

Solved) - The following answers were given by two students in response to a... (1 Answer) | Transtutors

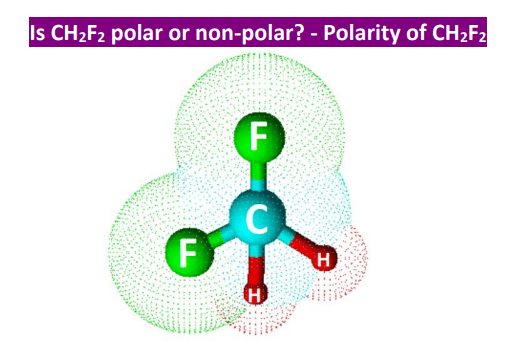
SOLVED: Why is CH2F2 polar while CH4 and CF4 are both non-polar? Please explain why this is the case. Thank you!

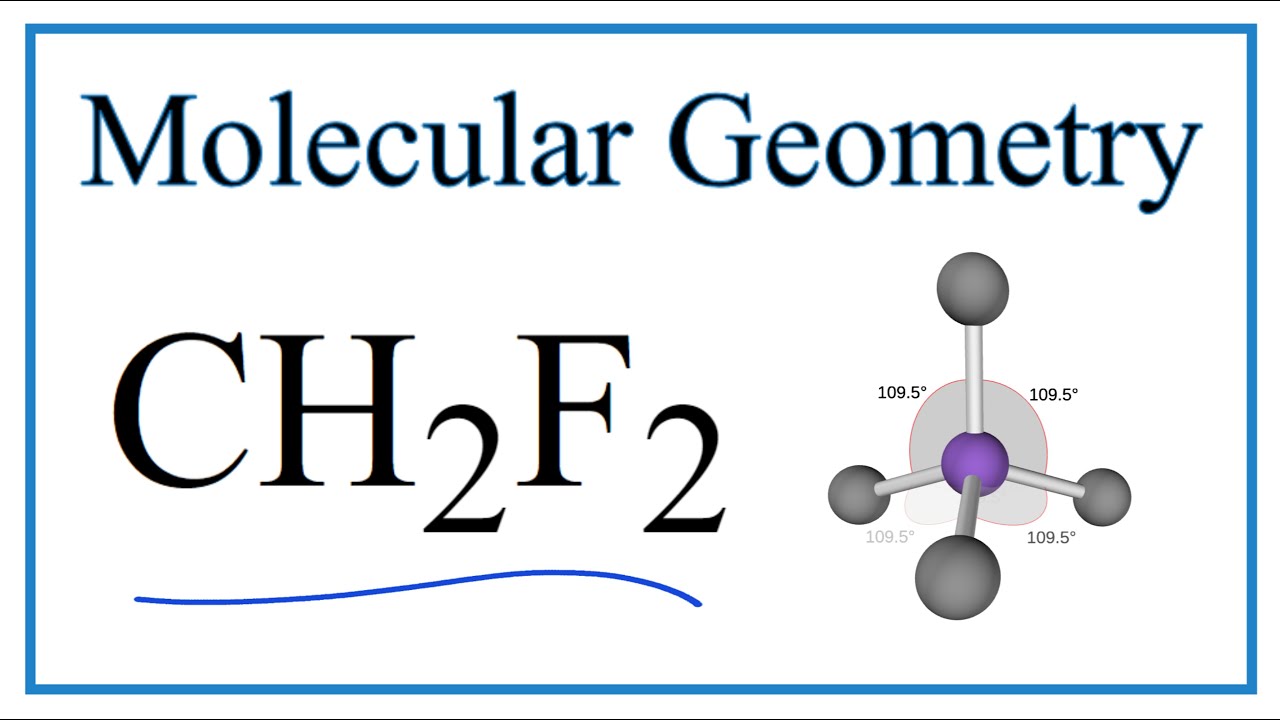
CH2F2 Lewis Structure (Difluoromethane) | CH2F2 Lewis Structure (Difluoromethane) This video will help you determine the CH2F2 Lewis structure with a step-by-step procedure. For finding the... | By Geometry Of Molecules